Organization of the Periodic Table:
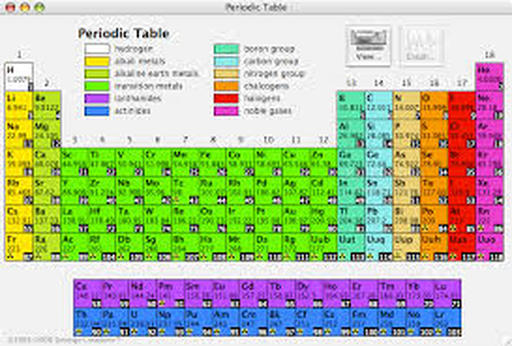
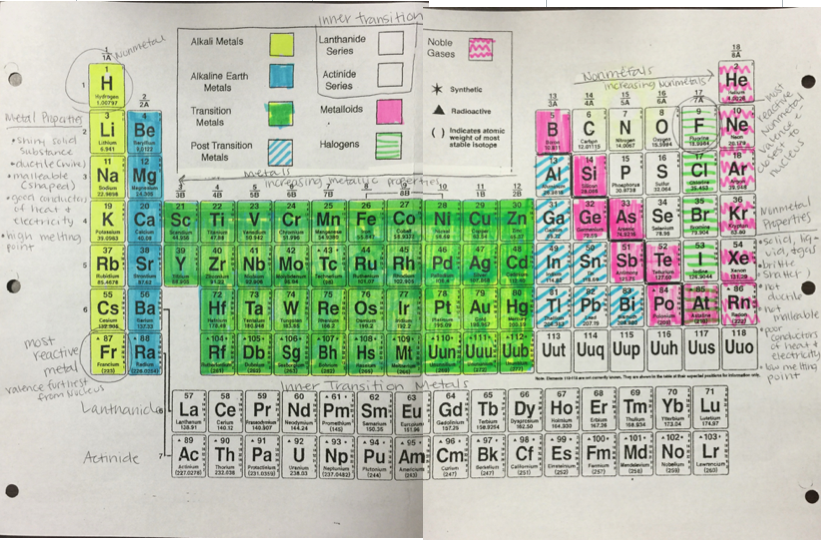
The periodic table organizes the elements in horizontal rows, or periods, by order of increasing atomic number, which equals the number of protons in the atomic nucleus of each element.
The periodic table is organized like a big grid. Each element is placed in a specific location because of its atomic structure. As with any grid, the periodic table has rows (left to right) and columns (up and down). Each row and column has specific characteristics. For example, beryllium (Be) and magnesium (Mg) are found in column two and share certain similarities while potassium (K) and calcium (Ca) from row four share different characteristics.
The periodic table is organized like a big grid. Each element is placed in a specific location because of its atomic structure. As with any grid, the periodic table has rows (left to right) and columns (up and down). Each row and column has specific characteristics. For example, beryllium (Be) and magnesium (Mg) are found in column two and share certain similarities while potassium (K) and calcium (Ca) from row four share different characteristics.
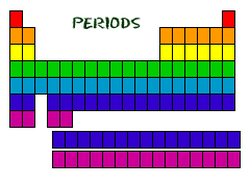
A period is a horizontal row <----> on the periodic table. You have a total of 7 periods. This number represents the number of energy levels an atom has.
For example: Hydrogen is in period 1, so it has 1 energy level. Potassium is in period 4, so it has four energy levels surrounding the nucleus.
Even though they skip some squares in between, all of the rows read left to right. When you look at the periodic table, each row is called a period (Get it? Like PERIODic table.). All of the elements in a period have the same number of atomic orbitals. For example, every element in the top row (the first period) has one orbital for its electrons. All of the elements in the second row (the second period) have two orbitals for their electrons. As you move down the table, every row adds an orbital. At this time, there is a maximum of seven electron orbitals
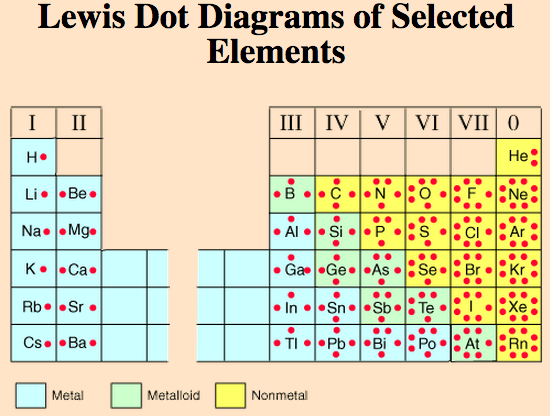
Now you know about periods going left to right. The periodic table also has a special name for its vertical columns. Each column is called a group. The elements in each group have the same number of electrons in the outer orbital. Those outer electrons are also called valence electrons. They are the electrons involved in chemical bonds with other elements.
A group, or also called a family, is a vertical column on the periodic table. There are a total of 18 columns on the periodic table. This number represents how many electrons are on the outer most energy level.
For example: Hydrogen is in group 1, therefore it has 1 electron on its outer most energy level. Calcium is in group 2, so it has two electrons on the outer most energy level.
A group, or also called a family, is a vertical column on the periodic table. There are a total of 18 columns on the periodic table. This number represents how many electrons are on the outer most energy level.
For example: Hydrogen is in group 1, therefore it has 1 electron on its outer most energy level. Calcium is in group 2, so it has two electrons on the outer most energy level.
The Basics:
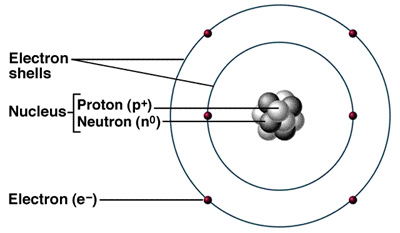
The atom is the basic structure of all materials. It contains protons, neutrons, and electrons. The protons and neutrons are in the center, called the nucleus. The electrons orbit around the nucleus on energy levels, or electron shells within the electron "cloud".
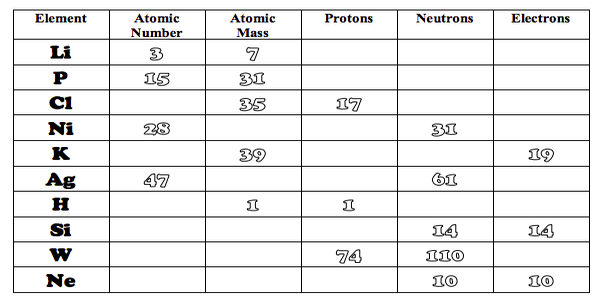
The atomic number on each element of the periodic table represents the number of protons AND electrons in the atom.
Protons AND Electrons
The atomic mass number represents the number of protons PLUS the number of electrons.
Neutrons + Protons = mass number
The atomic number on each element of the periodic table represents the number of protons AND electrons in the atom.
Protons AND Electrons
The atomic mass number represents the number of protons PLUS the number of electrons.
Neutrons + Protons = mass number
Electron Configuration:
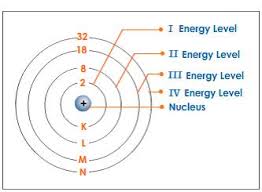
Each energy level on the periodic table can only hold a certain number of electrons.
On the outer most level of electrons, you will have your last energy level. This is called valence: the outer most level of electrons.
- 1st level/shell: 2 electrons
- 2nd level/shell: 8 electrons
- 3rd level/shell: 18 electrons
- 4th level/shell: 32 electrons
On the outer most level of electrons, you will have your last energy level. This is called valence: the outer most level of electrons.
Diagrams:
There are two different ways atoms can be drawn. One is to see how the atom arrangement is. This is the Bohr diagram. The other is mostly used for bonding purposes. This is called the Lewis-Dot structure.
The Bohr model is one of the most familiar models around. It takes into account the planetary model of an atom. In this model, the electrons are bound, or pulled, towards the positive center of the atom.
The Bohr model is one of the most familiar models around. It takes into account the planetary model of an atom. In this model, the electrons are bound, or pulled, towards the positive center of the atom.
Bonding
All elements attempt to reach one of the following:
This will make the atom stable, or "happy". Only one family on the periodic table is naturally stable, Noble Gases.
All other atoms have to bond in order to achieve this. Chemical compounds are formed by the joining of two or more atoms. A stable compound occurs when the total energy of the combination has lower energy than the separated atoms. The bound state implies a net attractive force between the atoms ... a chemical bond. The two extreme cases of chemical bonds are: Covalently or Ionically. Atoms will either share their valence electrons or transfer their valence electrons.
Covalent bond: bond in which one or more pairs of electrons are shared by two atoms.
Ionic bond: bond in which one or more electrons from one atom are removed and attached to another atom, resulting in positive and negative ions which attract each other.
Other types of bonds include metallic bonds and hydrogen bonding. The attractive forces between molecules in a liquid can be characterized as van der Waals bonds.
Think of it this way: if two elements on the periodic table are on opposite sides, such as Sodium and Chlorine, they can not share. One must give up an electron to another atom. If the elements
- a full outer shell
- 8 valence electrons
This will make the atom stable, or "happy". Only one family on the periodic table is naturally stable, Noble Gases.
All other atoms have to bond in order to achieve this. Chemical compounds are formed by the joining of two or more atoms. A stable compound occurs when the total energy of the combination has lower energy than the separated atoms. The bound state implies a net attractive force between the atoms ... a chemical bond. The two extreme cases of chemical bonds are: Covalently or Ionically. Atoms will either share their valence electrons or transfer their valence electrons.
Covalent bond: bond in which one or more pairs of electrons are shared by two atoms.
Ionic bond: bond in which one or more electrons from one atom are removed and attached to another atom, resulting in positive and negative ions which attract each other.
Other types of bonds include metallic bonds and hydrogen bonding. The attractive forces between molecules in a liquid can be characterized as van der Waals bonds.
Think of it this way: if two elements on the periodic table are on opposite sides, such as Sodium and Chlorine, they can not share. One must give up an electron to another atom. If the elements
Covalent Bonds:
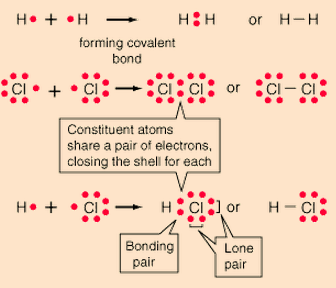
In covalent bonds, atoms share electrons. Covalent bonds form between two nonmetals.
Covalent chemical bonds involve the sharing of a pair of valence electrons by two atoms, in contrast to the transfer of electrons in ionic bonds. Such bonds lead to stable molecules if they share electrons in such a way as to create a noble gas configuration for each atom.
Hydrogen gas forms the simplest covalent bond in the diatomic hydrogen molecule. The halogens such as chlorine also exist as diatomic gases by forming covalent bonds. The nitrogen and oxygen which makes up the bulk of the atmosphere also exhibits covalent bonding in forming diatomic molecules.
In covalent bonds, atoms share electrons. Covalent bonds form between two nonmetals.
Covalent chemical bonds involve the sharing of a pair of valence electrons by two atoms, in contrast to the transfer of electrons in ionic bonds. Such bonds lead to stable molecules if they share electrons in such a way as to create a noble gas configuration for each atom.
Hydrogen gas forms the simplest covalent bond in the diatomic hydrogen molecule. The halogens such as chlorine also exist as diatomic gases by forming covalent bonds. The nitrogen and oxygen which makes up the bulk of the atmosphere also exhibits covalent bonding in forming diatomic molecules.
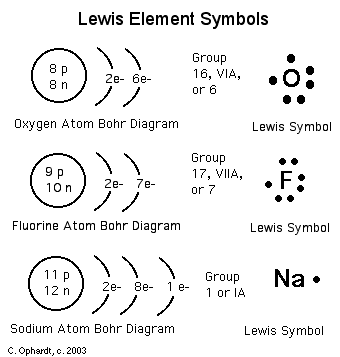
Covalent bonding can be visualized with the aid of Lewis Diagrams.
Ionic Bonds:
Ionic bonds form between a metal and a nonmetal. This is were they transfer electrons, causing the once neutral atom to now have an overall - or + charge. These are called ions. Ions can be either positively charged (+), called a cation, or negatively (-) charged, called an anion.
In ionic bonding, if the valence number is below 4, the element will lose all 4. If the valence is higher than 4, the element will gain the rest to reach 8, satisfying the octet rule.
Ionic bonds form between a metal and a nonmetal. This is were they transfer electrons, causing the once neutral atom to now have an overall - or + charge. These are called ions. Ions can be either positively charged (+), called a cation, or negatively (-) charged, called an anion.
In ionic bonding, if the valence number is below 4, the element will lose all 4. If the valence is higher than 4, the element will gain the rest to reach 8, satisfying the octet rule.
Comparing and Contrasting
Covalent & Ionic Bonds
Covalent & Ionic Bonds
Compounds VS Molecules
A compound is a substance that different atoms joined together by a covalent or ionic bonds.
A molecule is covalently bonded atoms which may contain one or more elements.
ALL compounds are molecules, but not all molecules are compounds!
A molecule is formed when two or more atoms join together chemically. A compound is a molecule that contains at least two different elements. All compounds are molecules but not all molecules are compounds.
Molecular hydrogen (H2), molecular oxygen (O2) and molecular nitrogen (N2) are not compounds because each is composed of a single element. Water (H2O), carbon dioxide (CO2) and methane (CH4) are compounds because each is made from more than one element. The smallest bit of each of these substances would be referred to as a molecule. For example, a single molecule of molecular hydrogen is made from two atoms of hydrogen while a single molecule of water is made from two atoms of hydrogen and one atom of oxygen.
A molecule is covalently bonded atoms which may contain one or more elements.
ALL compounds are molecules, but not all molecules are compounds!
A molecule is formed when two or more atoms join together chemically. A compound is a molecule that contains at least two different elements. All compounds are molecules but not all molecules are compounds.
Molecular hydrogen (H2), molecular oxygen (O2) and molecular nitrogen (N2) are not compounds because each is composed of a single element. Water (H2O), carbon dioxide (CO2) and methane (CH4) are compounds because each is made from more than one element. The smallest bit of each of these substances would be referred to as a molecule. For example, a single molecule of molecular hydrogen is made from two atoms of hydrogen while a single molecule of water is made from two atoms of hydrogen and one atom of oxygen.
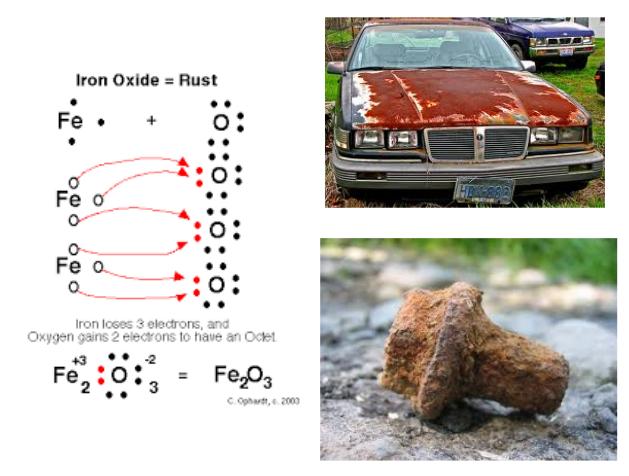
Let's look at Iron Oxide: Rust!
Balancing Equations
The law of conservation of mass states that in a chemical reaction the mass of the reactants equals the mass of the products. This means, when you have a certain number of atoms on one side of the yield symbol, you HAVE to have that same number on the other side.
pH of Substances
pH stands for the percent of Hydrogen atoms in a substance.
What does it mean for a solution to be acidic or basic (alkaline)?
It all has to do with hydrogen ions (abbreviated with the chemical symbol H+). In water (H2O), a small number of the molecules dissociate (split up). Some of the water molecules lose a hydrogen and become hydroxide ions (OH−). The "lost" hydrogen ions join up with water molecules to form hydronium ions (H3O+). For simplicity, hydronium ions are referred to as hydrogen ions H+. In pure water, there are an equal number of hydrogen ions and hydroxide ions. The solution is neither acidic or basic.
An acid is a substance that donates hydrogen ions. Because of this, when an acid is dissolved in water, the balance between hydrogen ions and hydroxide ions is shifted. Now there are more hydrogen ions than hydroxide ions in the solution. This kind of solution is acidic.
A base is a substance that accepts hydrogen ions. When a base is dissolved in water, the balance between hydrogen ions and hydroxide ions shifts the opposite way. Because the base "soaks up" hydrogen ions, the result is a solution with more hydroxide ions than hydrogen ions. This kind of solution is alkaline.
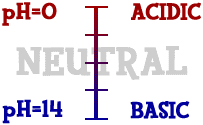
Scientists use something called the pH scale to measure how acidic or basic a liquid is. Although there may be many types of ions in a solution, pH focuses on concentrations of hydrogen ions (H+) and hydroxide ions (OH-). The scale measures values from 0 all the way up to 14.
Distilled water is 7 (right in the middle).
Acids are found between 0 and 7.
Bases are from 7 to 14.
Most of the liquids you find every day have a pH near 7. They are either a little below or a little above that mark. When you start looking at the pH of chemicals, the numbers can go to the extremes. If you ever go into a chemistry lab, you could find solutions with a pH of 1 and others with a pH of 14. There are also very strong acids with pH values below 1, such as battery acid. Bases with pH values near 14 include drain cleaner and sodium hydroxide (NaOH). Those chemicals are very dangerous.
What does it mean for a solution to be acidic or basic (alkaline)?
It all has to do with hydrogen ions (abbreviated with the chemical symbol H+). In water (H2O), a small number of the molecules dissociate (split up). Some of the water molecules lose a hydrogen and become hydroxide ions (OH−). The "lost" hydrogen ions join up with water molecules to form hydronium ions (H3O+). For simplicity, hydronium ions are referred to as hydrogen ions H+. In pure water, there are an equal number of hydrogen ions and hydroxide ions. The solution is neither acidic or basic.
An acid is a substance that donates hydrogen ions. Because of this, when an acid is dissolved in water, the balance between hydrogen ions and hydroxide ions is shifted. Now there are more hydrogen ions than hydroxide ions in the solution. This kind of solution is acidic.
A base is a substance that accepts hydrogen ions. When a base is dissolved in water, the balance between hydrogen ions and hydroxide ions shifts the opposite way. Because the base "soaks up" hydrogen ions, the result is a solution with more hydroxide ions than hydrogen ions. This kind of solution is alkaline.
Scientists use something called the pH scale to measure how acidic or basic a liquid is. Although there may be many types of ions in a solution, pH focuses on concentrations of hydrogen ions (H+) and hydroxide ions (OH-). The scale measures values from 0 all the way up to 14.
Distilled water is 7 (right in the middle).
Acids are found between 0 and 7.
Bases are from 7 to 14.
Most of the liquids you find every day have a pH near 7. They are either a little below or a little above that mark. When you start looking at the pH of chemicals, the numbers can go to the extremes. If you ever go into a chemistry lab, you could find solutions with a pH of 1 and others with a pH of 14. There are also very strong acids with pH values below 1, such as battery acid. Bases with pH values near 14 include drain cleaner and sodium hydroxide (NaOH). Those chemicals are very dangerous.